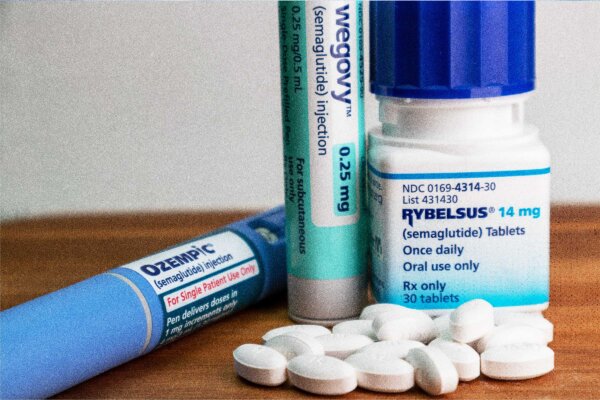
Health Canada has approved a second generic version of the diabetes drug Ozempic, marking the first such product produced by a Canadian pharmaceutical company.
The semaglutide injection submitted by Canadian drugmaker Apotex was authorized after a review found it met federal safety, efficacy, and quality standards, according to a Health Canada news release. The generic drug will be administered as a once-weekly injection, consistent with existing semaglutide treatments.
We had a problem loading this article. Please enable javascript or use a different browser. If the issue persists, please visit our help center.